Donald Trump has been in disagreement this week with top scientists by claiming that the anti-malarial drug chloroquine should be adopted immediately for treating patients with COVID-19, and even given as a prophylaxis. In this blog I will explore the challenges facing scientists working to develop drugs to treat COVID-19 and discuss recent advances, including chloroquine.
What are the challenges in designing anti-viral drugs? After all, there are a lot of antibiotics out there that work for a whole range of different bacterial infections. Let’s look at the structure of a bacterium, shown below.
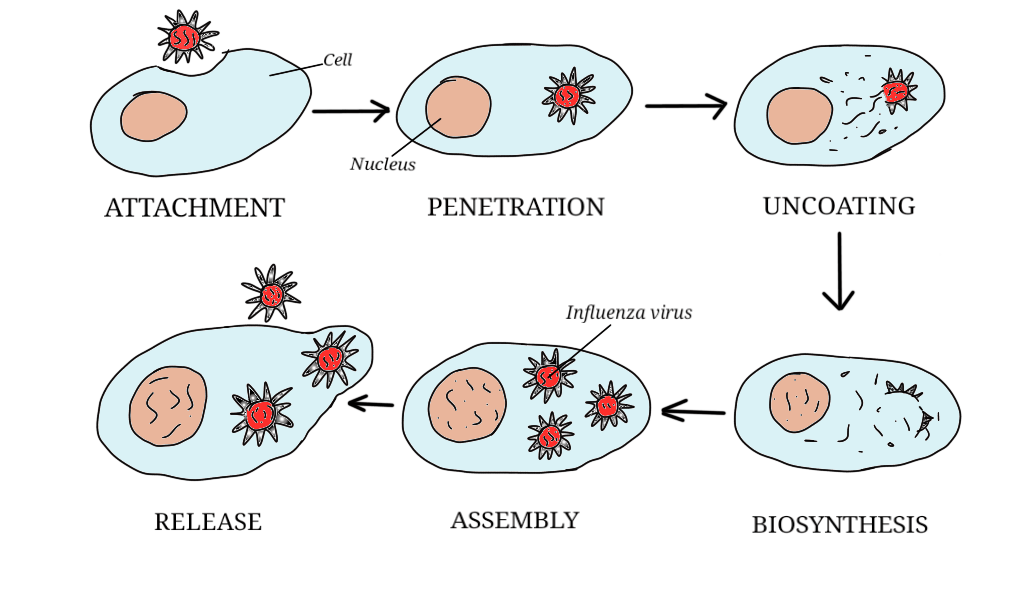
Compared with a virus, the structure of a bacterium, shown below, is highly complex. Bacteria carry out a huge number of chemical reactions and process. They release energy through respiration; they make proteins; they replicate their DNA in order to reproduce. And that’s just for starters; there are many, many more processes going on. Viruses, by contrast, can’t do any of these things without infecting a cell and using its organelles.
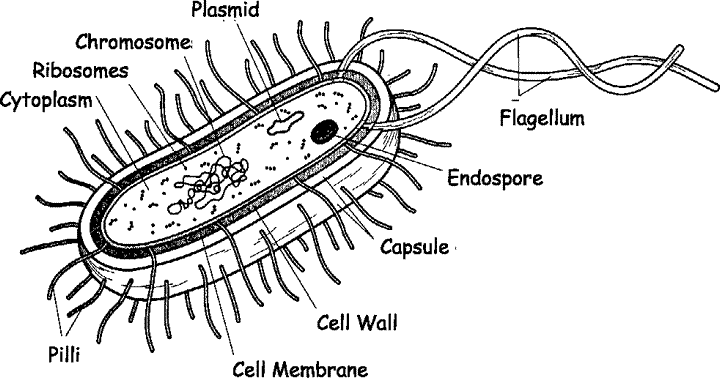
With bacteria, inhibiting or blocking even one of these reactions or processes can be enough to kill it. All chemical reactions in living organisms are controlled by enzymes, which are a type of protein. Each protein controls one, specific reaction. Disable the enzyme, and you stop the reaction.
For example, penicillin works by disabling the enzyme that bacteria use to maintain and repair their cell walls. The cell wall disintegrates, and the bacterium dies. Baytril, an antibiotic widely used in veterinary medicine, interferes with the replication of DNA, meaning that the bacterium can’t reproduce.
So, why are viruses harder to fight? The answer lies in the fact that viruses are so simple, meaning there are far fewer reactions and processes for drugs to target. One virus that is treated very successfully with drugs is HIV. So, why can’t some of the drugs used to treat HIV be adapted to use against COVID-19? By virus standards, HIV is quite complex. It’s a type of virus called a retrovirus, which has a more complicated reproductive cycle than other viruses. To put it simply, most of the drugs used to treat HIV are blocking or inhibiting processes that just don’t happen in COVID-19. Two HIV drugs, lopinavir and ritonavir, have shown some promise in treating COVID-19.
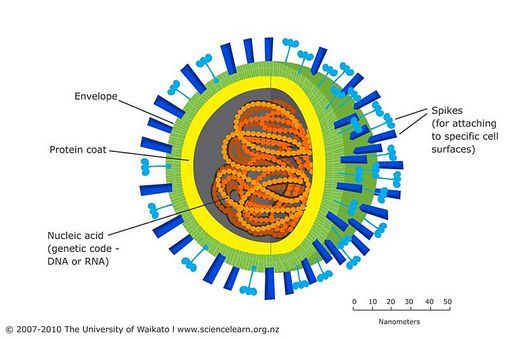
One of the most promising potential treatments is a drug called favipiravir. It’s a drug being developed in Japan, specifically to treat RNA viruses, and COVID-19 is an RNA virus. Originally developed as a treatment for influenza, trials have been carried out in China on 340 patients who had tested positive for COVID-19. It was found that the drug significantly shortened the time taken for patients to test negative, and that respiratory symptoms and lung function improved as well.
Why then, is favipiravir not being adopted immediately? The answer is that it has only been licensed to treat influenza. This means that extensive clinical trials must take place before it can be licensed for COVID-19 to ensure that it is safe and effective.
What about chloroquine? Chloroquine is an old drug, it’s been used for over 80 years to treat malaria, which is a disease caused by a parasite carried by mosquitos. It has been known for a while that chloroquine has antiviral activity. It basically prevents viruses from sticking to the membranes of cells, meaning they cannot enter and infect them.
So far, the evidence supporting chloroquine is anecdotal – this means it does not meet the standard to be considered reliable. Some doctors in China and South Korea have given it to patients with COVID-19 and seen improvements; and in France, 24 patients were given a similar drug, hydroxycloroquine, and showed improvements.
These were small scale experiments and not carried out under the controlled conditions of a clinical trial. As a result though, 6 properly designed clinical trials are getting underway around the world to test whether chloroquine or hydroxychloroquine are safe and effective against COVID-19.
If either chloroquine or hydroxychloroquine do turn out to be effective, it would be a major breakthrough. Because both have been used for many years in treating malaria, they are already known to be safe. They are also safe to take during pregnancy; there are concerns that favipiravir may have adverse effects on a developing foetus. Both drugs are cheap, and facilities to manufacture them in large amounts are already up and running.
There are downsides. The side effects of chloroquine or hydroxychloroquine are pretty unpleasant, mainly nausea and diarrhoea. In rare cases chloroquine can cause blindness. An important consideration too is their use in treating malaria. Drug resistance in malaria is a big concern to those involved in treating it.
There are already a number of strains of malaria that are resistant to both chloroquine or hydroxychloroquine. The risk malaria resistance must be evaluated before using them to treat COVID-19 in places where malaria is active. For example, it’s now malaria season in southern Africa and malaria will be active until around the end of April. In India, malaria season begins in May.
In terms of drug development, there is a lot to be optimistic about. It may well be that an effective treatment is developed before a vaccine. Certainly, many scientists are now beginning to talk about a timescale of months, rather than years, for developing an effective treatment. I will keep you all up to date about advances as they occur, meanwhile, stay safe and well!