One of the most frequently asked questions at the moment is how long it will take to develop an effective vaccine for coronavirus COVID-19. An effective and safe vaccine would undoubtedly be the best way to halt the spread of the virus. There’s some excellent news on that front – the first human testing of a vaccine began in the US earlier this week. This is an incredible achievement to have got to human testing within such a short time! In this post I’ll explain some of the challenges facing vaccine developers, and some of the amazing leaps forward that have been made in recent weeks.

Edward Jenner is credited with the discovery of vaccination, but in fact, a crude (and highly risky) form of immunisation against smallpox, called variolation, had been in existence for centuries. Variolation dates back to 10th century China, and the earliest form involved taking a powder made of crushed smallpox scabs and then blowing it up children’s noses with a tube – a practice called nasal insufflation. Yes, revolting I know! In Sudan, mothers of babies with smallpox would sell the scabs to other mothers who would rub them on the skin of their children.
By the time Jenner made his discoveries, variolation had been common practice in England for around 80 years and usually involved making a cut or scratch on the arm and introducing pus taken from infected patients. Yes, still revolting! The problem with variolation is that it involved deliberately infecting someone with a dangerous disease. The idea was that variolation gave a much milder illness, and after recovery, the variolated person was immune. The risk was kept to a minimum by ensuring that the material used in variolation was taken only from patients with mild illness; nevertheless, a small but significant proportion of those variolated would get the full-blown disease, leading to disfigurement or even death.

I won’t go into Jenner’s story in detail here, as it is covered in plenty of other places. In short, Jenner discovered that innoculating someone with cowpox, which is a similar virus to smallpox but a very mild illness, gave immunity to smallpox.
Early vaccines were developed basically using trial and error. A major breakthrough was made by Louis Pasteur, who discovered that bacteria and viruses could be artificially weakened, and that the weakened strains induced immunity with either mild symptoms or no symptoms. Pasteur’s arch-rival Robert Koch pioneered the use of dead pathogens (bacteria and viruses) as vaccines; later, the discovery of antibodies enabled better understanding of how vaccines work.
There have been cases where a vaccine has been developed and produced very rapidly. In 1957, a new strain of influenza emerged which had the potential to become a global pandemic. In 9 days, American microbiologist Maurice Hilleman had isolated the new virus, allowing development of a vaccine and saving many thousands of lives.

This begs the question, why can’t a COVID-19 vaccine be developed that quickly? After all, the virus has been isolated and its genome sequenced. A major difference is that Hilleman was working with a virus that was already well understood – it was simply a new strain. Vaccinations for flu use either dead virus (adults) or live, weakened virus (children). The manufacturing process basically involves growing the required strain in chicken eggs. It’s reliable and can be used for any strain of flu once the virus has been isolated.
A second point is that in 1957, regulatory processes were nothing like as strict as they are now. Hilleman’s Hong Kong flu vaccine did not have to go through the extensive stages of development, testing and approval that a modern vaccine is subjected to before it reaches clinical use. Even so, for a well-understood virus like flu, a vaccine can make it into clinical use relatively quickly. In April 2009, the first cases of a new strain of H1N1 swine flu were reported; by November a vaccine was in clinical use.
So, what are the challenges of developing a vaccine from scratch? Vaccines make use of antigens. These are proteins on the surface of a pathogen that signal the immune system to attack it. As part of the immune response, antibodies will be made to match the antigen. Your immune system then ‘remembers’ how to make those antibodies, so that if you encounter the pathogen again, you can fight it off before symptoms develop.
A major challenge is identifying which type of vaccine will be most effective. Inactivated vaccines use a dead pathogen; the pathogen is killed by heat, radiation or chemical treatment, then introduced into the body to provoke an immune response. Attenuated vaccines use a weakened form of the pathogen. They are highly effective but cannot be given to people with compromised immune systems. Protein sub-unit vaccines use either a protein from the surface of a virus, or the virus envelope with the DNA or RNA removed. DNA or RNA vaccines use part of the virus’ DNA/ RNA; cells in the body take this up and make proteins that mimic those that the virus produces; provoking an immune response.
It’s also important to make sure a vaccine is resilient to small mutations of the virus. This is especially important because COVID-19 is an RNA virus and they mutate much more easily and often than DNA viruses – see my post about how viruses change. These mutations are very small, and modern vaccine developers have the tools to create vaccines that will still be effective if the virus undergoes small changes.
A COVID-19 vaccine will need to be 3 things: safe, effective and resilient to minor mutations of the virus. Vaccine development has two stages: pre-clinical and clinical. In the pre-clinical phase, scientists study the virus and its antigens; they test it in test tubes and on animal models; and they develop the manufacturing process. In the clinical phase, there are a series of carefully structured and monitored trials.
So, where are we with COVID-19? A major development is that scientists have determined the structure of the spike protein on the surface of the virus, which acts as an antigen. More about how this was done, and why it’s important, in a separate post.
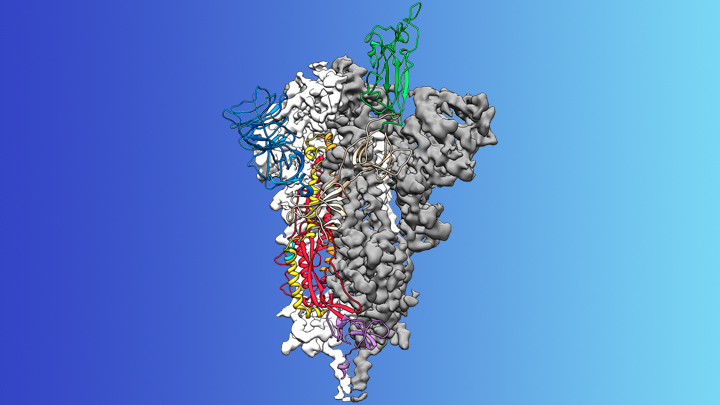
In brief, studying the structure of the spike protein in detail will help vaccine developers create that mutation resilience I talked about earlier, because they can ensure that a vaccine will still be effective if there are changes to the spike protein. In addition, now the spike protein has been fully characterised, it’s possible to manufacture it artificially and also manufacture artificial antibodies; scientists can use both of these in vaccine research, which is easier and safer than using the actual virus.
Another important tool is genome sequencing, which I explained in an earlier post. Again, in brief, scientists all over the world are sequencing the genome of the virus and entering results into a massive international database. This allows them to track changes in the RNA of the virus, which is also very important in ensuring that a vaccine is resilient to minor changes.
Earlier this week, a vaccine in the USA entered what is known as Phase I clinical trials – this is when the vaccine is tested on a small number of carefully selected, healthy volunteers to assess safety and immune response. If those are successful, Phase II trials will begin; these involve a larger group of volunteers who will be vaccinated and then deliberately infected with the vaccine. Phase III would be a large scale trial involving hundreds of volunteers in multiple locations to test whether the vaccine is safe and effective across the population as a whole. If that is successful, the manufacturers can apply for a license and the vaccine can be adopted for clinical use.
I will stress here, only a small proportion of Phase I trials progress to Phase II. However, to have a vaccine in Phase 1 trials this quickly (within about 3 months of the virus appearing) is amazing progress. I’m keeping a very close eye on vaccine development and will keep you all updated, so watch this space!