Haber’s life was the tragedy of the German Jew – the tragedy of unrequited love – Albert Einstein
Almost exactly 106 years ago, on the afternoon of April 22nd 1915, things were fairly quiet on the Northern edge of the Ypres Salient between Gravenstafel and Langemark. Possibly too quiet, as subsequent events would demonstrate. But for the French Territorial and Colonial troops in the trenches, there was nothing to indicate the horror that was about to be unleashed.

As the afternoon drew on, a slight breeze began to blow from the direction of the German lines. At approximately 5pm, a strange greenish-grey mist was observed moving towards the Allied lines from the German trenches. It was chlorine gas, and when it reached the French trenches, all hell broke loose. Men died in agony as the chlorine reacted with moisture in the linings of their airways to form corrosive hydrochloric acid. The French troops broke and ran, leaving 4 miles of the front line completely undefended. The Germans could have broken through to Ypres and on to the Channel Ports; however, two things saved the day for the Allies.
The first was that the effectiveness of the gas took even the Germans by surprise, and they simply did not have enough troops to exploit the breakthrough. The second was the almost unbelievable courage of the Canadian troops on the Southern flank of the breakthrough. They moved into the trenches the French had abandoned and fought off multiple gas attacks. In two days, the Allies lost about 5000 killed and 15000 wounded. It was the first large-scale use of a lethal, asphyxiating gas in warfare, and set the scene for 3 years of horror as both sides raced to develop ever more effective chemical weapons.

The man behind it all was a quiet, bespectacled, middle-aged German army lieutenant named Fritz Haber, who would become one of the most controversial scientists of the 20th century.
Before the War, Haber had been a Professor of chemistry at the University of Karlsruhe, and in 1911 had become the first director of the new Kaiser Wilhelm Institute in Berlin. His most important work, however, was carried out while he was at Karlsruhe.
Look at a graph of world population, like the one shown below, and you will see that until about 1800 ACE, population growth was very slow. From 1800 on, the world’s population began to grow with increasing rapidity, as improvements in food production, medical care and living conditions began to reduce mortality rates. As the end of the 19th century approached, the world’s population was heading towards unsustainable levels. Quite simply, the human population was about to exceed the Earth’s capacity to produce food.
The problem was a shortage of nitrogen. This element is essential for plant growth, and without plants and their ability to photosynthesise, there is no food. But nitrogen is one of the most abundant elements on Earth, so how could there be a shortage? The answer is that the bulk of the Earth’s nitrogen is in the atmosphere, where it cannot directly be used by plants. Plants require water-soluble nitrates, which can be taken up from the soil by their roots.
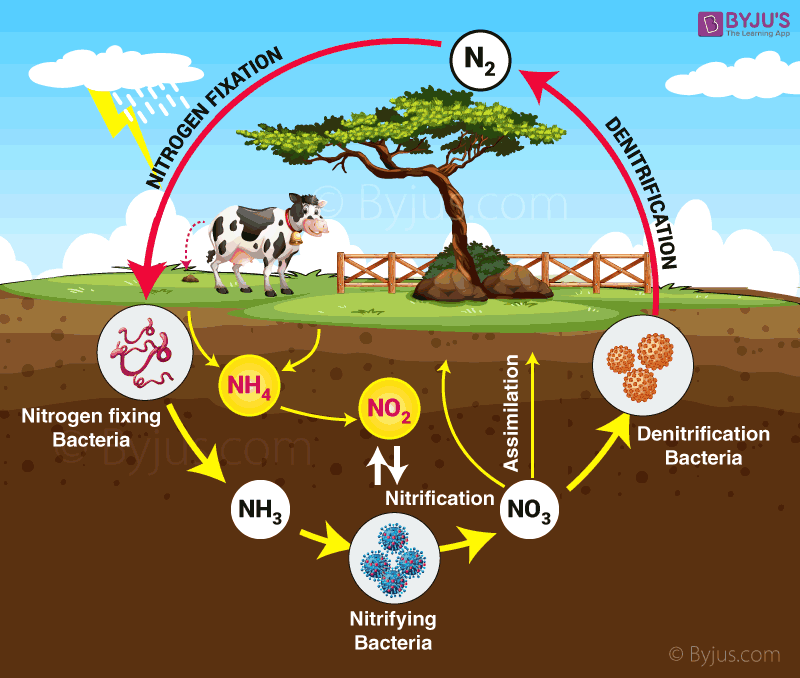
In the ordinary way of things, this is taken care of by Mother Nature. Bacteria in the soil and in the root nodules of leguminous plants ‘fix’ nitrogen into ammonia, and a small amount of atmospheric nitrogen is also ‘fixed’ by lightning. Other bacteria and fungi convert nitrogen from dead plant and animal matter into ammonia; a third group of bacteria then convert the ammonia into nitrates. This is known as the nitrogen cycle. By the late 19th century, these natural nitrogen-fixing processes could no longer produce enough nitrates to meet the increasing need for food crops. A cull of the world’s population by means of a catastrophic world-wide famine was imminent, unless a way could be found of artificially fixing atmospheric nitrogen.

Enter Fritz Haber and his assistant, Robert Le Rossignol, who in the summer of 1909 succesfully produced ammonia by reacting hydrogen and nitrogen gases at high temperature and pressure, using an iron catalyst. Once you have ammonia, it is then relatively straightforward to convert it into nitrates for fertilisers.

Carl Bosch of the BASF company scaled-up the process to industrial level, with the first operational ammonia plant opening in 1913 at Oppau. Now known as the Haber-Bosch process, this was one of the biggest breakthroughs industrial chemistry has ever seen, and paved the way for the large-scale manufacture of nitrate fertilisers, meaning that food production could continue to increase and meet the needs of the expanding population.

By a (not so?) happy coincidence, Haber and Bosch’s work also meant that if war broke out, Germany would be able to manufacture nitrates to make explosives. Prior to the invention of the Haber-Bosch process, the main source of nitrates for both fertilisers and explosives was guano. Yes, you did read that right, guano as in bird poo. Guano is a rich source of nitrates, and the best sources of guano in terms of both quality and quantity could be found in South America, particularly the Chincha Islands in Peru, where millions of seabirds create mountains of guano up to 150 feet high. In the 19th century, the importation of guano by sea was big business. In the event of a war, it was possible, even likely, that a Royal Navy blockade would be able to prevent Germany from importing guano. The Haber-Bosch process meant that in this event, Germany’s munitions factories could continue to churn out shells and bullets. Despite this, the invention of ammonia synthesis was, overall, something that benefited humankind. So how did Haber go from that to chemical warfare?
Fritz Haber was intensely patriotic. Part of this was due to his having been born into a Jewish family. In the early 20th century, antisemitism in Germany was nowhere near as prevalent as it would become in the Nazi era. However, it was significant enough for Haber to feel that despite his having converted to Christianity, he needed to prove his patriotism and devotion to the Fatherland. Haber was enthusiastic about the war, and was one of 93 German intellectuals to sign a proclamation which enthusiastically endorsed the declaration of war.

In addition to his patriotism, Haber had two areas of expertise that made him the ideal person to lead Germany’s efforts to develop poisonous gas as a weapon. The first was in electrochemistry, which is crucial in the manufacture of chlorine gas. The second was in working with gases under pressure, which enabled him to develop equipment with which to deliver the gas, and meant that throughout the war, German equipment was superior to anything used by the allies.
For the duration of the War, Haber led the German efforts to develop both chemical weapons and defences such as gas masks. The result was that Germany was always one step ahead of the allies in both. It is estimated that there were about 1.3 million casualties as a result of gas, with approximately 90,000 fatalities – relatively trivial in relation to the overall numbers of casualties. But gas was, first and foremost, a weapon of terror; its aim was to disrupt, damage morale and consume resources treating the wounded. During the first gas attack at Second Ypres, many of the troops who broke and ran did so out of sheer terror, having seen the effects of the gas on others. Mustard gas in particular was designed to maim and terrorise rather than kill.

Haber’s involvement in chemical warfare was to cost him dearly. On 2nd May 1915, his wife Clara committed suicide, shooting herself through the heart with Haber’s own army revolver while her husband was celebrating his promotion to captain. A dedicated pacifist and a talented chemist in her own right, her husband’s involvement in the war is thought to have been a major factor in Clara’s suicide.
After the war, Haber came in for a great deal of criticism from the scientific community. He was awarded the 1918 Nobel Prize for Chemistry for his work on ammonia synthesis, and this was controversial to put it mildly. Many eminent scientists boycotted the award ceremony in 1919, and it is said that Sir Ernest Rutherford refused to shake Haber’s hand.
In the post-war years, Haber continued as Director of the Kaiser Wilhelm Institute, where he led Germany’s secret efforts to develop new chemical weapons, in direct contravention of the Treaty of Versailles. But dark clouds were gathering on the horizon. When Hitler became Chancellor in 1933, he immediately began to target Jewish scientists. Haber was personally targeted, with Hitler claiming that his appointment as director of the Institute was a result of his being the nephew of a Jewish profiteer (which he wasn’t). This came as a shock to Haber, who had thought that his service in WW1 and his conversion to Christianity would be enough to protect him. Although Haber could not legally be dismissed, the Nazis made his position untenable and he resigned in October 1933. He left Germany and after a brief spell in England, in January 1934 he set out to take up a new job in Palestine. But he never reached his destination. Events had taken a severe toll on Haber’s health and on 29th January 1934 he died of heart failure while breaking his journey in Switzerland.

There is a final hideous twist to the story of Fritz Haber, the scientist hounded out of Nazi Germany because of his Jewish roots. Between the wars, the research Haber was overseeing at the Kaiser Wilhelm Institute included the development of new pesticides and fumigants. This included the development of a highly effective class of fumigants which consisted of hydrogen cyanide gas adsorbed onto diatomaceous earths. When exposed to air, the lethal gas was released. These agents were given the name ‘Zyklons’ and included Zyklon B, which would be used in the murder of 1.1 million people in the Holocaust. Among those who would perish in the gas chambers were several members of Fritz Haber’s own extended family.
