In the fourth post in my series on how scientists study viruses, I am going to talk about crystallography. Crystallography involves shining a beam of X-rays through a crystal. By looking at how the X-rays pass through the crystal, scientists can actually generate a detailed picture of the structure of a molecule and how the atoms are arranged.
So, how does it work? Think of the waves you see in the ocean. They are straight lines, more or less, like the picture on the left. Now think of what happens when you throw a stone into a pond. The waves spread out in a circle from where the stone hit the water, as in the picture on the right.

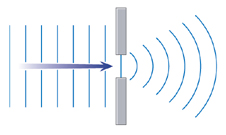
Now, let’s look at what happens when a straight wave is forced through a narrow gap. It spreads out in a half-circle. This is called diffraction and is shown in the diagram on the right. What does this have to do with studying viruses? It’s to do with the fact that X-rays behave like waves.
Basically, if you shine X-rays through a crystal, tiny gaps between the atoms act like the narrow gap in the diagram and make the waves spread out into circles. These then meet waves emerging from other gaps, causing an interference pattern. You can see an interference pattern for yourself by dropping two stones into water close together, and watching what happens when the waves meet. By analysing the interference patterns when X-rays pass through a crystal, scientists can work out the arrangements of the atoms.

One of the most important scientists in the development of X-ray crystallography was Dorothy Crowfoot Hodgkin. She solved the structures of cholesterol and penicillin, and in 1956 she found the structure of vitamin B12, for which she was awarded the Nobel Prize in 1964. However, she is best known for solving the structure of insulin in 1969.
The basic principals of crystallography are no different today to what they were in 1914 when the first crystal structure (table salt) was solved. What has revolutionised crystallography is the development of computers.
To get from measurements of interference patterns to a molecular structure requires some truly horrendous maths – one of the few bits of my chemistry degree that I really didn’t enjoy at all! It took Dorothy Hodgkin 30 years to solve the structure of insulin, and insulin is a comparatively simple molecule. Modern computer processing means that scientists can determine the structures of very complex molecules, like the spike protein on the surface of COVID-19, relatively quickly.
So, how are scientists using crystallography to study COVID-19? They are using it to determine the chemical structures of molecules that make up the virus. The most important of these is possibly the spike protein. This is on the surface of the virus, and the virus uses it to attach itself to the surface of a human cell. An important part of how your immune system fights the virus is by making antibodies which are exactly the right shape to attach to the spike protein.
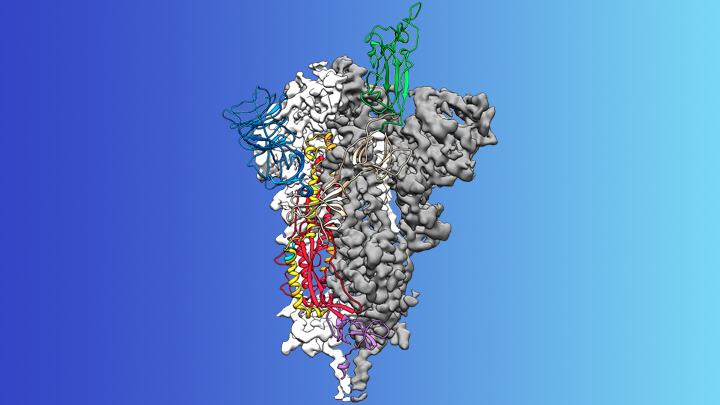
Scientists have been able to isolate the spike protein and grow a pure crystal; they have then used crystallography to solve the structure. Knowing the exact structure of the spike protein will help those developing vaccines and anti-viral drugs to combat COVID-19. Also, scientists can monitor how the spike protein changes when the virus mutates; this knowledge will help to make sure that any vaccine will continue to work if there are mutations.
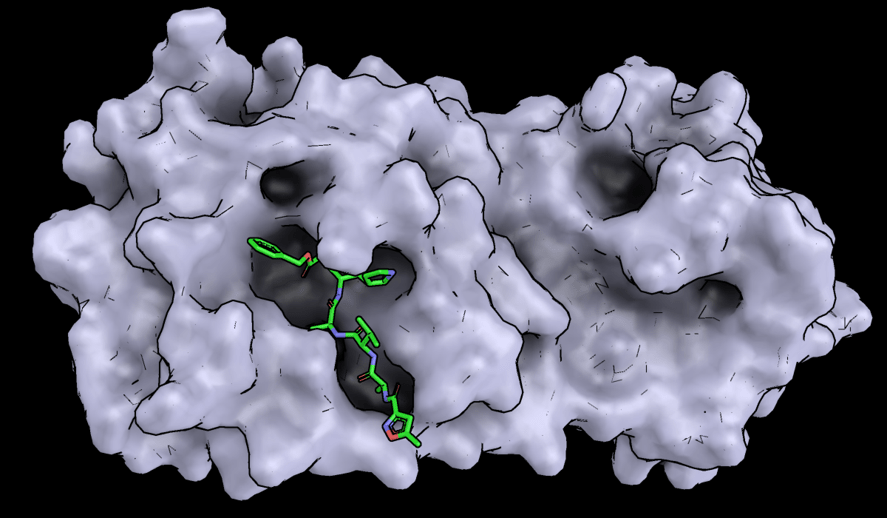
The structures of other important molecules in the virus have also been solved, including an enzyme called protease. Enzymes are substances that control chemical reactions; protease controls the production of the proteins needed to make new viruses. By solving the structure, scientists have identified how it could be stopped from working. As a result, a team in Shanghai are investigating 30 (yes, 30!) molecules that could be used to treat COVID-19. And that’s just one of hundreds of teams around the world.
I hope this has been interesting/ helpful. As a scientist myself, I am in awe of what has been achieved in such a small time, and how much we now know about the virus. There is actually plenty of good news out there, if you know where to look for it, and you can be sure that I will keep you updated of new discoveries as and when they happen. In fact, I’ve just found out that some drug trials in China are showing real promise, so watch out for a post about that later today.